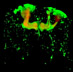
The brainbow
Professor Jeff Lichtman describes the events leading to his team's development of the 'brainbow,' a new technique for staining cells.
My colleagues and I had been eager to come up with a strategy to see the connections of the nervous system, and the problem is that the nervous system itself is kind of bland. The nerve cells themselves don’t have any pigments in them to see. If you look at a piece of nervous system on a slide, or look into a whole brain, you just see a milky mass of nothing that you can discern. What humans have done for a long time is stain nervous systems with various kinds of labels to allow one to see cells. The problem with the nervous system is that there are billions of cells, and they are all smushed in together so tightly that if you stain them all you’re back to where you started from. Instead of being a white mass of stuff you don’t understand now maybe it would be green, but it would all be green, or it would all be red. What we were thinking about, and I thought about this for many years related to the way video monitors on a computer works or the way a color television works is that with a few pigments like red, green, and blue one can get a wide range of colors by mixing various ratios of those colors. So on your computer monitor wherever you see a yellow area, in fact there’s no yellow pixel in the circuitry of your screen of your computer, there are just green and red pixels right next to each other and green plus red looks to the human eye as yellow. So what we decided to do was see if we could make mice in which individual nerve cells had random combinations of three genes, each of which encode for a different color fluorescent protein. One of these fluorescent proteins is red, another is blue, and another is green, RGB. So if you had a different amount of the R plus the G plus the B in each cell, each cell would have it’s own color. That was the hope and John Livet, a very hardworking and amazingly intuitive molecular biologist, came up with a couple of strategies to try to do this, and with my colleague Josh Sanes we worked for about five years and developed mice now that have a random mixture of three fluorescent proteins in each nervous system cell and these cells now come in about 150 colors. The brains are quite beautiful to look at because every cell has its own unique shade. This allows us to trace out all the wires in a small region in a way that was not possible with any other techniques. My main role in this was, besides helping with the imaging, was to come up with a good name and I realized that these were brains and they sort of looked like rainbows and we kept talking about rainbow brains and finally 'brainbow' just popped up and that name stuck.
brainbow, color, colour, pigments, rgb, stain, staining, microscopy, fluorescent, fluorescence, jeff, lichtman
- ID: 2063
- Source: DNALC.G2C
- Download: MPEG 4 Video Windows Media Video Theora Video
Related Content
2053. What is fluorescence microscopy?
Professor Jeff Lichtman introduces fluorescence microscopy, a powerful technique of illuminating minuscule molecules for analysis by very powerful microscopes.
2054. Fluorescence microscopy - pros and cons
Professor Jeff Lichtman examines the technique of fluorescence microscopy in terms of its benefits (e.g. exquisite resolution) and its drawbacks (e.g. confined by the wavelength of light).
2055. Spatial resolution and neuroimaging
Professor Jeff Lichtman discusses spatial resolution in relation to a number of imaging techniques including MRI, fluorescence microscopy, and electron microscopy.
2061. Microscopes, nanoscopes, and finer resolution
Professor Jeff Lichtman examines the development of imaging technologies from the days of Cajal to the development of the nanoscope.
864. Imaging Technology
Images from brain scans and new microscopy techniques are offering a strikingly clear glimpse of what’s going on underneath the bumpy surface of our skulls.
15562. Human chromosomes / karyotype (colored)
A portrait of human chromosomes: this process labels the chromosomes with multicolored fluorescent tags, allowing researchers to consistently distinguish between chromosomes.
15527. DNA microarray
DNA microarray
16119. DNA sequencing, Leroy Hood
Leroy Hood talks about DNA sequencing.
2048. Dynamic imaging and fluorescent microscopy
Professor Rusty Lansford explains that dynamic imaging is important because it allows researchers to examine active development rather than interpreting a series of snapshots.
1721. 3D Gene Expression
Like all brains, insect brains have different structures that accomplish specific tasks. Dr. Josh Dubnau introduces a technique for examining gene expression in the brains of fruit flies.