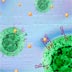
Pathways, Releasing the protein
In this section learn that newly made proteins leave the endoplasmic reticulum wrapped in a layer of membrane called a vesicle.
The newly made proteins leave the endoplasmic reticulum wrapped in a layer of membrane called a vesicle. They travel toward the Golgi apparatus (on the right) where the proteins are modified and sorted for transport. The Golgi is busy with protein traffic moving in and out. The vesicle fuses with the membrane at one end of the Golgi and a new vesicle containing the modified proteins is pinched off the other side. The proteins are transported through the cytoplasm and delivered to where they are needed. Some proteins are used inside the cell. Others, like these growth factors, must be exported to function. The vesicle fuses with the cell membrane, dumping the proteins outside the cell. The released proteins will signal surrounding cells, or, in some pathways to cancer, will coax this cell into further action. Molecules identified: Growth factor: Proteins that stimulate cell division, proliferation or differentiation. See EGF and PDGF. Platelet derived growth factor (PDGF) receptors: PDGF receptors are large membrane-spanning proteins with an extracellular and an intracellular component. Two PDGF receptor proteins "dimerize" to bind a single platelet-derived growth factor. The cytoplasmic portion has kinase activity – able to add phosphate molecules to other molecules to activate them. This receptor can contribute to cancer if rendered active for an extended period of time. Ras: A protein loosely associated with the inner surface of the cell membrane. The Ras protein binds guanine nucleotides – guanosine diphosphate (GDP) and guanosine triphosphate (GTP). When a stimulatory signal arrives, Ras releases its GDP and acquires a GTP molecule, entering an active state and emitting a signal to another protein. After transmitting the signal, Ras deactivates itself by cleaving a phosphate molecule from the GTP to reduce it to GDP, or another protein called Ras-GAP comes in to break the GTP down. A single amino acid change can alter the function of Ras, causing it to bind GTP but making it unable to deactivate. Ras mutations are prevalent in many human cancers, including colon, skin, and lung.
protein traffic, receptor proteins, ras protein, guanosine triphosphate, pdgf receptor, amino acid change, platelet derived growth factor, vesicle, golgi apparatus, diphosphate, active state, inner surface, gtp, cell membrane, cytoplasm, growth factors, nucleotides, extracellular, receptors
- ID: 1024
- Source: DNALC.IC
Related Content
1019. Pathways, Beneath the membrane
In this section learn that the binding of growth factors outside the cell causes receptors ends to intertwine and activate each other, and once active, the modified receptor ends interact with messenger proteins.
1020. Pathways, A bevy of interactions
In this section learn that receptors activate each other before binding an adaptor molecule and an exchange factor.
1018. Pathways, At the cell surface
In this section learn that a signaling pathway begins with the arrival of a chemical signal – such as a hormone or growth factor – at the cell surface.
16877. Cell Signals
Journey inside a cell as you follow proteins and learn about cellular interactions. This 3-D animation brings to life the inner workings of a fibroblast cell as it responds to external signals. Created by Cold Spring Harbor Laboratory and Interactive Know
959. Causes, Smoking: K-ras
This section explains how the protein produced by the K-ras gene is a tumor “activator.”
16724. Concept 35: DNA responds to signals from outside the cell.
Signal transduction is cell communication that involves a series of molecular transformations.
1023. Pathways, Making the protein
In this section learn that in the cytoplasm, the messenger RNA is released from its carrier proteins and binds to a protein assembly complex called a ribosome.
868. Candidate Genes for Schizophrenia
An interactive chromosome map of the genes and loci associated with schizophrenia.
1022. Pathways, Inside the nucleus
In this section learn that an activated protein is transported into the nucleus through a pore in the nuclear membrane.
1069. Receptor Molecules
Professor David Van Vactor describes the role of receptor molecules, which receive signals from outside the cell, passing the signal to the inside.